
News - AI-Powered Insight Reveals Brain Rewiring After Stroke
Business Strategy
AI-Powered Insight Reveals Brain Rewiring After Stroke
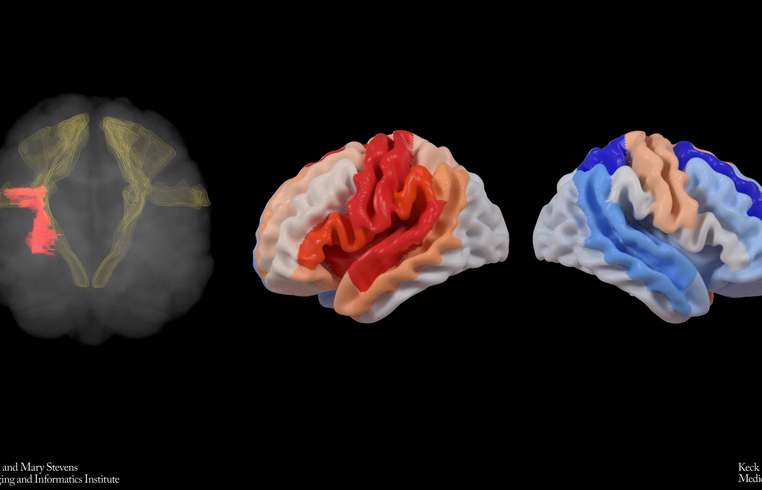
In groundbreaking research highlighted in The Lancet Digital Health, scientists from the USC Mark and Mary Stevens Neuroimaging and Informatics Institute (Stevens INI) have discovered a phenomenon where stroke-affected brains may reorganize themselves, reflecting structurally youthful features in undamaged regions to adapt post-injury. Initiated by the Enhancing NeuroImaging Genetics through Meta-Analysis (ENIGMA) Stroke Recovery Working Group, the international study explored brain scans of over 500 stroke survivors from 34 research facilities across eight different nations. By utilizing deep learning models trained on a vast array of MRI scans, the team assessed the 'brain age' of various brain regions post-stroke to examine structural impact and recovery patterns. Dr. Hosung Kim, associate professor of research neurology at the Keck School of Medicine of USC, noted, "The occurrence of larger strokes tends to age the affected hemisphere of the brain, yet intriguingly, it may render the opposite side to appear younger. This observation suggests a compensatory brain adaptation, rejuvenating viable networks to regain lost functionality." Using a sophisticated AI graph convolutional network, the team medically assessed 18 distinct brain regions from MRI data to determine the biological age. Discrepancies between these predictions and actual ages, known as brain-predicted age difference (brain-PAD), serve as indicators of neural health and recovery. Pairing these metrics with motor performance scores unveiled an astounding finding: Even at six months post-rehabilitation, stroke survivors with significant movement challenges displayed a younger-than-expected brain age within regions opposite to the lesion, particularly within the frontoparietal network — crucial for motor functions. "Our research signifies that severe stroke aftermath may prompt healthy brain sections, particularly those contrary to the lesion, to compensate for movement deficits," Dr. Kim elaborated. "The contralesional frontoparietal network, a determinant in motor planning and coordination, portrayed rejuvenated characteristics, indicating neural contingency rather than complete functional recovery. This dimension of neuroplasticity had eluded traditional imaging technologies until now." This pivotal research, conducted under ENIGMA, aggregates neural data globally to enrich our understanding of brain dynamics across various ailments. MRI data alignment and clinical metrics from diverse cohorts culminated in composing the world's largest stroke neuroimaging collection. Arthur W. Toga, director of the Stevens INI, emphasized, "Pooling global stroke survivor data with AI allows us to detect intricate brain reorganization patterns that smaller studies might miss. These diverse aging patterns in chronic post-stroke brains could pioneer personalized rehabilitation strategies." Plans are underway to extend this study longitudinally, tracking the evolution of brain age and reorganization from acute stages through stroke recovery chronologies. Clinicians envision patient-specific interventions designed around individual neural adaptations, potentially enhancing recovery prospects swiftly. The study titled “Deep learning prediction of MRI-based regional brain age reveals contralesional neuroplasticity associated with severe motor impairment in chronic stroke: A worldwide ENIGMA study,” was funded by the NIH grant R01 NS115845, engaging international partners from the University of British Columbia, Monash University, Emory University, and the University of Oslo. For more details on this study or related ENIGMA research progressions, visit https://enigma.ini.usc.edu.






